Introduction:
On this page you can learn how to classify elements from the periodic table. You can also learn the difference between periods and groups, some history of the periodic table, and all of the group names (providing examples and uses).
On this page you can learn how to classify elements from the periodic table. You can also learn the difference between periods and groups, some history of the periodic table, and all of the group names (providing examples and uses).
Periodic Table
The tabular arrangement of the elements according to their atomic numbers so that elements with similar properties are in the same column.
Concepts:
Classifying Elements
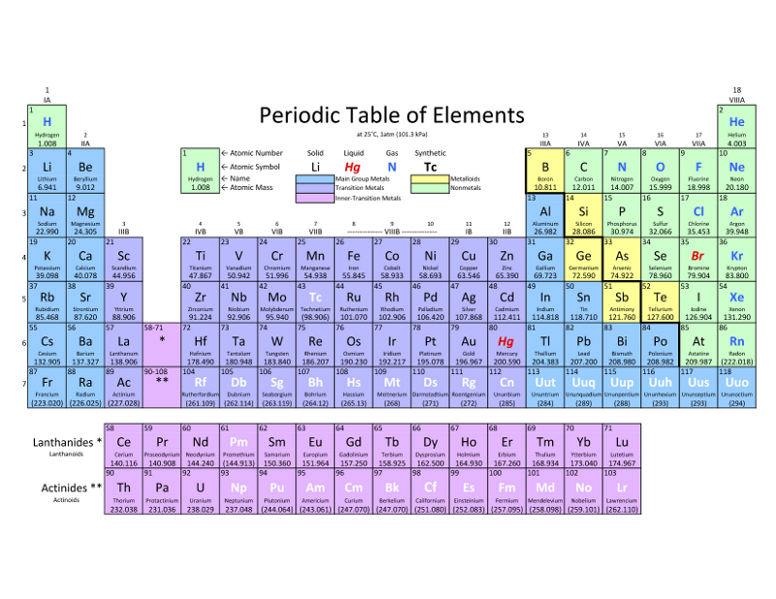
The periodic table arranges all of the known elements in order of increasing atomic number. Order generally coincides with increasing atomic mass.
Periods Vs. Groups
The different rows of elements are called periods. The period number of an element signifies the highest energy level an electron in that element occupies (in the unexcited state). The number of electrons in a period increases as one moves down the periodic table; therefore, as the energy level of the atom increases, the number of energy sub-levels per energy level increases. Elements that lie in the same column on the periodic table (called a "group") have identical valance electron configurations and are chemically similar. For instance, all the group 18 elements are inert, or noble gases.
History
Dimitri Mendeleev arranged the elements in his periodic table in order of increasing atomic mass. His periodic table is similar to what we use today.
Lother Meyer published a nerly identical table but Mendeleev got more credit for publishing first.
Classification
The 3 classifications of elements are metals(the majority of the periodic table), nonmetals(upper right corner), and metalloids(heavy stair-step line that separates metals from non metals).
metals- good conductors of heat and electric current.
nonmetals- poor conductors of heat and electric current.
metalloids- have properties similar to metals and nonmetals.
Group Names
Alkali metals: elements in group 1A(rich in sodium and potassium)
Alkaline Earth metals: elements in group 2A
Halogens: nonmetals in group 7A(contain salts)
Transitional metals: elements in group B(usually displaid in the main body of the periodic table)
Noble Gasses: elements in group 8A( nonmetals, sometimes called inert gasses because they rarely take place in reactions)
Inner Transitional metals: elements that appear below the main body of the periodic table(some are made in laboratories)
Artifacts:
[link]
Supporting Info:
periodic table
http://www.instagrok.com/results.html?query=periodic%20table
Connecting Topics:
Classifying Elements
The periodic table arranges all of the known elements in order of increasing atomic number. Order generally coincides with increasing atomic mass.
Periods Vs. Groups
The different rows of elements are called periods. The period number of an element signifies the highest energy level an electron in that element occupies (in the unexcited state). The number of electrons in a period increases as one moves down the periodic table; therefore, as the energy level of the atom increases, the number of energy sub-levels per energy level increases. Elements that lie in the same column on the periodic table (called a "group") have identical valance electron configurations and are chemically similar. For instance, all the group 18 elements are inert, or noble gases.
History
Dimitri Mendeleev arranged the elements in his periodic table in order of increasing atomic mass. His periodic table is similar to what we use today.
Lother Meyer published a nerly identical table but Mendeleev got more credit for publishing first.
Classification
The 3 classifications of elements are metals(the majority of the periodic table), nonmetals(upper right corner), and metalloids(heavy stair-step line that separates metals from non metals).
metals- good conductors of heat and electric current.
nonmetals- poor conductors of heat and electric current.
metalloids- have properties similar to metals and nonmetals.
Group Names
Alkali metals: elements in group 1A(rich in sodium and potassium)
Alkaline Earth metals: elements in group 2A
Halogens: nonmetals in group 7A(contain salts)
Transitional metals: elements in group B(usually displaid in the main body of the periodic table)
Noble Gasses: elements in group 8A( nonmetals, sometimes called inert gasses because they rarely take place in reactions)
Inner Transitional metals: elements that appear below the main body of the periodic table(some are made in laboratories)
Artifacts:
[link]
Supporting Info:
periodic table
http://www.instagrok.com/results.html?query=periodic%20table
Connecting Topics:
The periodic table is related to organic chemistry and atomic structures because it is the periodic table of elements which is the base of all chemistry.